Amir ASADIRAD
Ph.D., P.CHEM.

Life is full of challenges and science is full of unknowns. I challenge the life’s challenges and fight to know the unknowns.
My research interests lie at the interface of organic chemistry and materials science with focus on designing and synthesizing new photoresponsive materials such as molecular switches. These molecules change their structures and functions when triggered with light, electricity or chemical stimuli.
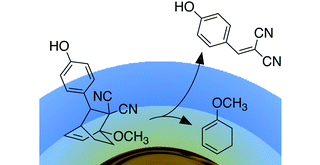
My goal is to integrate the photoresponsive molecules into drug delivery systems and use light as a trigger to selectively unmask small molecules for potential therapeutic applications.
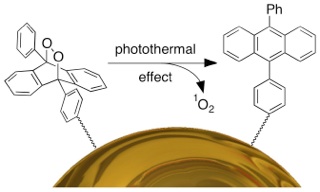
In my early studies, I have introduced the concept of releasing singlet oxygen as a therapeutic agent from the surface of gold nanoparticles (here). The exposure of nanoparticles to light generates heat during the photothermal process and consequently triggers the singlet oxygen release. This new proof-of-concept study can open a new highway to improve the efficiency of photodynamic therapy in future. This study is highlighted in the Chemistry World News (here).
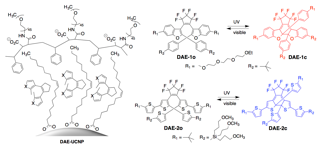
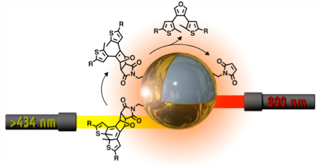
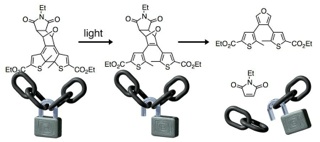
More recently, I have demonstrated that small molecules can be released from the surface nanoparticles using two colours of light and photoresponsive dithienylethene ligands (here). In this proof-of-concept study, I have demonstrated that the photothermal effect is not able to break bonds and release small molecules from the surface of SiO2–Au core–shell nanoparticles unless the nanosystem is first exposed to visible light. Similar to AND logic gate, this system requires both inputs (i.e NIR and visible light) to be present to achieve the derired output. This study represents a general and versatile approach to potentially address the limitations of technologies suchas drug delivery nanosysmtes.
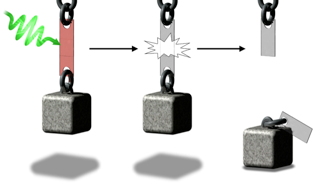
I also demonstrated that the photochromic dithienylethene derivatives are useful organic molecules for controlling the properties of macromolecules (here). The integration of these molecular switches into the polymer backbone allows the end-user to control the properties of polymer ‘on-command’. In this study, I have synthesized a thermally self-healing polymer by the Diels-Alder reaction between dithienylfuran and maleimide monomers. I have demonstrated that the exposure of the polymer to UV and Visible light ‘gate’ the reversibility of the Diels-Alder reaction and turn the self-healing properties of the polymer ‘off’ and ‘on’, respectively. After exposure to UV light, the strength of the polymer as an adhesive is enhanced. Whereas, the visible light weakens the adhesive.
Vancouver, BC
Burnaby, BC
Oceaneos Environmental Solutions
Vancouver, BC
Simon Fraser University, Burnaby, BC
Organic & Nanomaterials Science
Simon Fraser University, Burnaby, BC
Organic chemistry
Simon Fraser University, Burnaby, BC
Textile Chemistry and Fibre Science
Arak University, Iran
Technical Skills
Air & moisture Sensitive Reactions Organometallic Reactions Lithium-Halogen Exchange Cross-coupling Reactions Photochemical Syntehesis Pilot-scale Synthesis
AuNPs AuNRs SiO2NPs SiO2-Au Core-Shell NPs UCNPs
Schlenk Line Glove Box Degassing Cannula
Column Chromatography Crystallization Vacuum Distillation Extraction TLC Titration
(operation & data analysis)
FTIR 1H NMR (1D & 2D) 13C NMR Fluorescence Uv-vis
HPLC GPC LC-MS GC-MS ICP-MS
TEM SEM SEM-EDX DMA TGA PSA XRD DLS
In the past few years, I have taken many interesting photos of exciting reactions as well as many microscopy images. Some are listed below:
If you should need my assistance with your research, please do not hesitate to contact me.